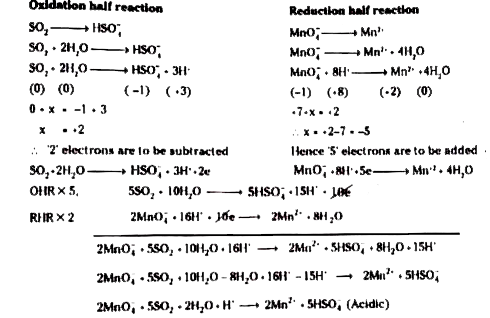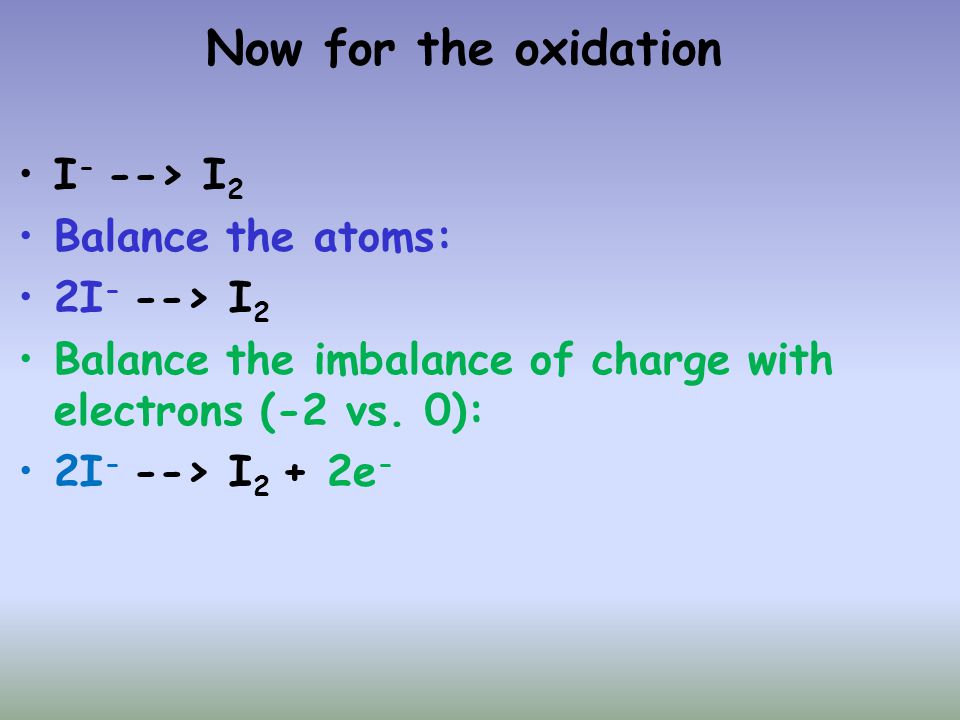SOLVED: 4. Write balanced equations for the following redox reactions:a. NaBr + Cl2 NaCl + Br2b. Fe2O3 + CO Fe + CO2 in acidic solutionc. CO + I2O5 CO2 + I2 in

Balance the following redox reactions by the ion - electron method in basis medium MnO^-4(aq) + I^-(aq)→ MnO2(s) + I2(s)

Hasbro Beyblade Burst - Evolution Single Pack - Ifritor I2 (Balance) - Right Spin Battle Top - Ages 8+ : Amazon.com.au: Toys & Games

MnO4 + I = MnO2 + I2 balance this equation by oxidation method in basic medium and give all the steps - Brainly.in

SOLVED: Iodide ion reacts with Manganese (IV) oxide according to the following balanced oxidation reduction equation: MnO2(s) + 2 I-(aq) + 4 H+(aq) → I2(aq) + Mn2+(aq) + 2 H2O(l) Which answer